Electron domain molecular geometry table12/2/2023  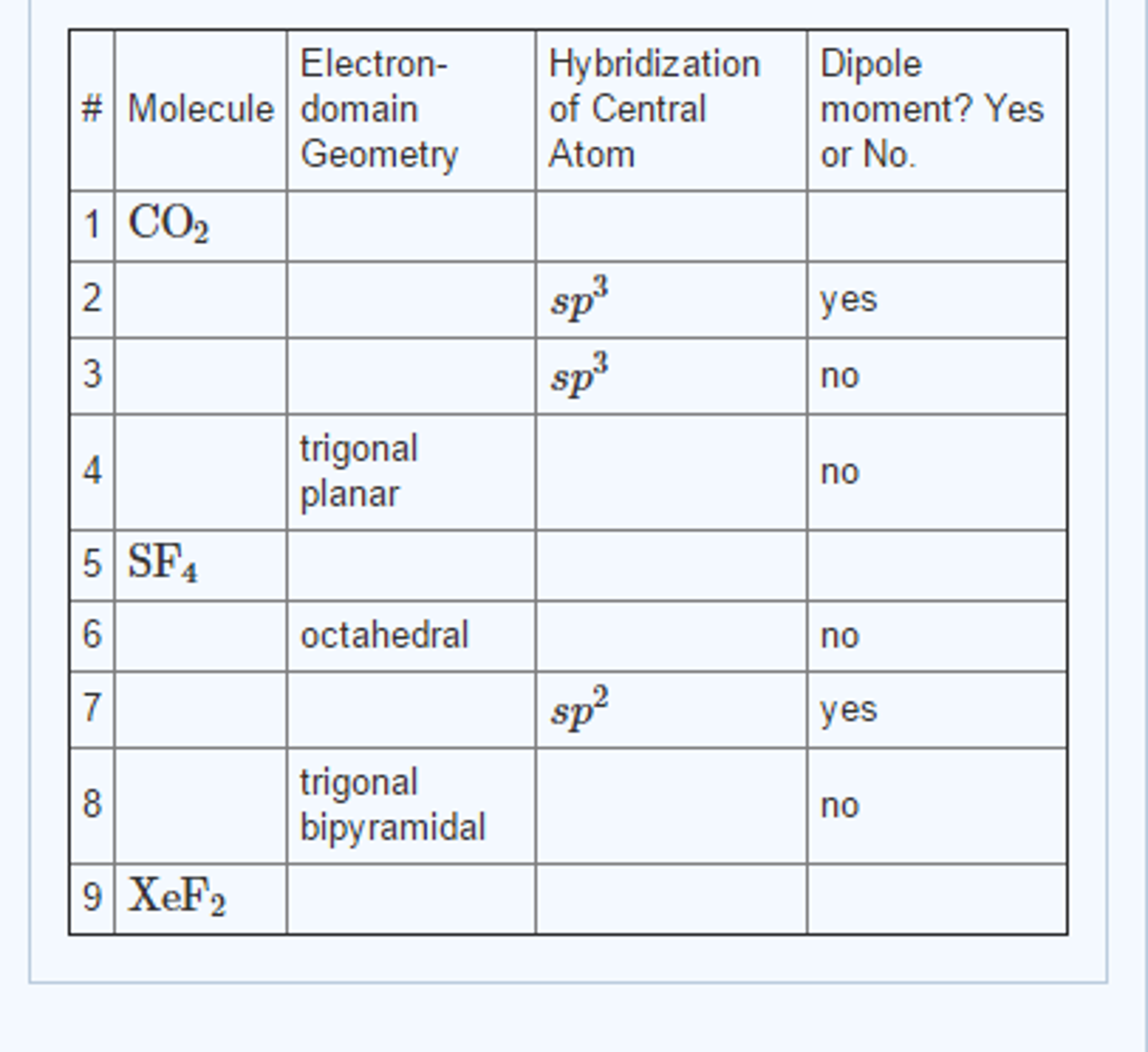
To predict the molecular geometry select from the table below the 3D arrangement that has the same number of bond domainsand lone pairs of electrons.ġ. Determine the number of bond domains and the number of lone pairs of electrons.ģ. Halogens and noble gases can expand their octet.Ģ. Note: To chose the central atom as the one with the smallest number of valence electrons or if they all have the same number of valence electrons then choose the one in the least amount. Draw Lewis structures for the molecular formula given. How to use the table to predict molecular geometry.ġ. 
For example, the molecule ethylene, H 2C=CH 2, has the carbon-carbon atoms sharing four electrons but one bond domain between the two carbon atoms. In any case, if two atoms share two electrons or more they will have one bond domain. 
For ease of understanding we will call a bond domain the electrons that hold together two atoms. The following table will help you understand how molecular geometry can be predicted using the VSPER model. On the other hand it takes into account the very important Pauli exclusion principle where each electron pair must occupy a different spatial region about an atom. On the first hand it minimizes repulsion between electrons due to electrostatic interactions. The VSPER model is based on two important principles. This model proposes that electrons are arranged around atoms in pairs such that they are kept as far away as possible. The valence shell electron pair repulsion (VSPER pronounced "vesper") model provides some useful tools for predicting molecular geometries. Atoms have a definite three-dimensional spacearrangement relative to each other in a molecule. can also be observed in the microscopic world. So it has four pairs altogether, giving it a tetrahedral arrangement and bent geometry (2-2 combination of 2 BP and 2 NBP), which corresponds to a 105 degree bond angle.The geometrical arrangements seen in nature, i.e., flowers,stones, trees, etc. Therefore, it has two bonding pairs of electrons (to each H) and also has 2 pairs of lone pairs (pair of dots). When drawing the Lewis structure of water, oxygen is the central atom. Double and triple bonds still count as 1 bonding domain (BD)!Įxample: H 2O.So count only the # pairs around the central atom not in the entire compound! NOTE: the # of BD and NBD is from the perspective of the CENTRAL ATOM.Now within that arrangement, what is the specific combination and geometry.Count the TOTAL number of electron domains (both bonding and non-bonding) and classify the ARRANGMENT.Draw the Lewis structure of the molecule.Each particular combination will determine a geometry.ĭetermining Electron Arrangement/Geometry Rules: Then we look to see exactly how the total electron domains are distributed-meaning how many are bonding and how many are non-bonding (or lone pairs). The total count will determine the arrangement. Electron domains are pairs of electrons so we count both bonding pairs and non-bonding pairs to get the TOTAL. We count the TOTAL # of electron domains. Arrangement is the general classification of “shape” but geometry is the specific “shape” of the molecule in question.įor example: 3 sided figures: triangles (general shape) BUT 3 sided figure with equal sides: equilateral triangle (a specific kind of triangle). Note: The difference between an arrangement and geometry is as follows. It allows us to predict the shape of a molecule 3-dimensionally. VSEPR: Valence Shell Electron Pair Repulsion model uses the repulsion of electron pairs to determine strain on various bonds and the resulting electron ARRANGEMENT and GEOMETRY. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
